I was frying potatoes over a campfire when I noticed the production of a green flame. Directly, the flame tests of the elements from chemistry classes came to my mind and I tried recognizing the source.
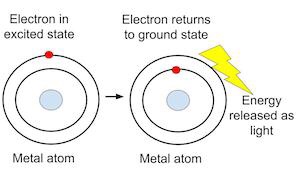
First, let us go over the flame test of chemicals: According to the qualitative analysis of chemical metal ions, the flame test visually determines the identity of an unknown metal ion according to the color of the flame. The heat of the flame excites the electrons of the metals ions into higher energy levels where they are energetically unstable. Hence, they tend to fall back down to their original energy levels, releasing energy as they do so in the form of light energy. This light has characteristic flame colors of different metal ions due to varying electron transitions.

As revealed from the flame test chart, boron ion B3+ and cupric ion Cu 2+ flame test give green flame. But where, how and why are these found in my woodfire?
At that day, I was burning in addition to wooden- logs the leaves of corns and most probably these were treated by a pesticide that has these chemicals as an active ingredients.


What are the compounds that may be found in pesticides and leading the green flame?
According to NPIC ( National pesticide Information Center): “Boric acid and its sodium borate salts are active ingredients in pesticides used against insects, spiders, mites, algae, molds, fungi, and weeds.” and as we know Boric acid B(OH) 3 is a compound of boron, oxygen, and hydrogen. This justifies the possible presence of boron ion in the woodfire.
Adding to this, according to the Best Management Handbook : “Copper pesticides are a group of many different compounds that have some form of copper in common as the active ingredient. These compounds have protectant activity against several bacterial and fungal diseases”. Again, this justifies the presence of cupric ions in the woodfire.
Next time, I have to reconsider burning the corn leaves due to the possible toxicity level of the released flame. Read more about the toxicity level of pesticides in Pesticide toxicity: a mechanistic approach.


Love it!
LikeLiked by 1 person
Interesting, and disturbing. American soil is saturated in pesticides.
LikeLiked by 1 person
Yes, indeed! The point that made me feel sad is that all of us push our kids to eat vegetables considering them healthy food!
LikeLike
Wonderful transition from observation to scary cause.
LikeLiked by 1 person